
Your Tactical Guide to Achieving PIC/S GMP Compliance for Global Market Access
In the global pharmaceutical market, compliance transcends mere regulatory adherence; it plays a pivotal role in facilitating international trade. A
We Embody a Legacy of Incredible Discoveries, Exploring the Boundaries of Innovation and Science

At Zenovel, we integrate our extensive expertise across a wide range of domains, from Good Clinical Practice to Regulatory Affairs, to present valuable insights and thought leadership. Each blog reflects our intense dedication to innovation, offering a forum to share our perspectives, groundbreaking ideas, and industry expertise. Through our blogs, we hope to uplift, inform, and support scientific, medical, and pharmaceutical innovation.

In the global pharmaceutical market, compliance transcends mere regulatory adherence; it plays a pivotal role in facilitating international trade. A

The pharmaceutical industry is facing a convergence of innovation and regulation as it approaches 2026, with GxP services playing a

The recent update of ICH E6(R3) marks a significant evolution in clinical trial execution, transitioning from a compliance-centric approach to
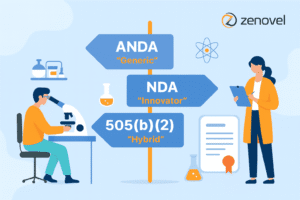
In the pharmaceutical industry, particularly in India’s expanding market, the regulatory pathway is a crucial determinant that influences the transition

In the current pharmaceutical environment, the integrity of a company’s supply chain relies heavily on its weakest link, particularly concerning
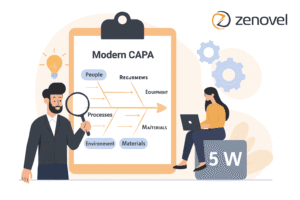
In clinical research, Good Clinical Practice (GCP) sets ethical and quality standards. Non-compliance during audits poses systemic risks to patient

Clinical trials’ credibility relies on strict compliance with Good Clinical Practice (GCP) guidelines, established by the International Council for Harmonisation (ICH). Zenovel offers expert GCP auditing services to help organizations maintain compliance, streamline operations, and prepare for regulatory inspections; ensuring clinical trials are always inspection-ready. Why GCP Audits Are Essential?

In the pharmaceutical manufacturing industry, a robust Quality Management System (QMS) is crucial for product safety, regulatory compliance, and operational excellence. Zenovel Pharma Services LLP, a global leader in clinical research and regulatory support, offers affordable, tailored services that support high standards of quality and efficiency. Our QMS setups are

Pharmacovigilance (PV), the scientific process of detection, assessment, understanding, and prevention of adverse drug reactions, is going to be revolutionize in 2025. PV is evolving from a more reactive to a proactive discipline, aided by new and evolving technologies, including quantum-enhanced AI, global safety monitoring in real time, and other

In the world of globalized pharmaceutical industry, clinical trials and regulatory affairs are becoming ever more international in terms of languages and cultures. India is well on the way to becoming a global pharmaceutical hub in 2025, now is the time to ensure that we get accurate translation and localization

The Indian pharmaceutical industry, a global leader and the third-largest medicine producer by volume, is renowned for providing cost-effective, high-quality drugs worldwide. In 2025, new regulatory frameworks and stringent compliance requirements are being implemented, compelling Indian pharma companies to adapt. These reforms aim to enhance Pharma Regulations credibility, safety, and

Steps to Optimize Your Regulatory Affairs Process Regulatory affairs professionals in medical technology are crucial in ensuring compliance and market entry. Such professionals face challenges in navigating registration processes, fostering collaboration, and utilizing data-driven insights. Innovative regulatory solutions can enhance efficiency and improve outcomes. This blog presents five transformative strategies
We provide affordable, innovative, and high-quality solutions to pharma industry with ethics driven research and increasing accessibility to high quality medicine worldwide
Copyright © 2024 Zenovel. All rights reserved.