
CPHI Milan 2026
CPHI Milan 2026 Schedule a Meeting With Our Experts For: GCP Audit/Monitoring GMP Audits & Consulting End to End Regulatory
We Embody a Legacy of Incredible Discoveries, Exploring the Boundaries of Innovation and Science

At Zenovel, we integrate our extensive expertise across a wide range of domains, from Good Clinical Practice to Regulatory Affairs, to present valuable insights and thought leadership. Each blog reflects our intense dedication to innovation, offering a forum to share our perspectives, groundbreaking ideas, and industry expertise. Through our blogs, we hope to uplift, inform, and support scientific, medical, and pharmaceutical innovation.

CPHI Milan 2026 Schedule a Meeting With Our Experts For: GCP Audit/Monitoring GMP Audits & Consulting End to End Regulatory

BioChina 2026 Schedule a Meeting With Our Experts For: GCP Audit/Monitoring GMP Audits & Consulting End to End Regulatory

CPHI Middle East 2026 Schedule a Meeting With Our Experts For: GCP Audit/Monitoring GMP Audits & Consulting End to

CPHI Korea 2026 Schedule a Meeting With Our Experts For: GCP Audit/Monitoring GMP Audits & Consulting End to End

CPHI & PMEC China 2026 Schedule a Meeting With Our Experts For: GCP Audit/Monitoring GMP Audits & Consulting End

iPHEX 2026 Schedule a Meeting With Our Experts For: GCP Audit/Monitoring GMP Audits & Consulting End to End Regulatory

The pharmaceutical industry is heavily regulated, and compliance with strict standards is crucial for operational excellence and avoiding penalties. To achieve regulatory readiness, pharmaceutical quality leaders use proactive preparation methods like mock inspections and compliance gap analysis. Zenovel guides companies through these processes to strengthen quality systems, enhance staff readiness,

Quality and compliance are paramount in the life sciences and pharmaceutical industry. Regulatory agencies like the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and the World Health Organization (WHO) have established rigorous requirements that rely on a holistic approach to quality assurance (QA) and validation. At Zenovel,

The pharmaceutical industry is growing and changing rapidly. As a result of this emergence, getting a new drug to the market can be a thorny and long path from the moment of research and development to the point of regulatory approval. At every step in that journey, carefulness, compliance, and

In the ever-changing and competitive pharmaceutical and biotechnology industry, long-lasting success requires strategic foresight, flexibility, and a firm grasp of the market environment. At Zenovel, we specialize in offering expert advisory services and integrated service solutions to help organizations navigate complex environments and achieve sustained services provided to clients. Below

Clinical trials are the backbone of pharmaceutical innovation; ensuring new treatments are safe and effective. However, managing these trials is no small feat—complex data, regulatory compliance, and tight timelines demand robust solutions. Clinical Trial Management System (CTMS), specialized software designed to streamline trial processes, enhance data accuracy, and improve operational
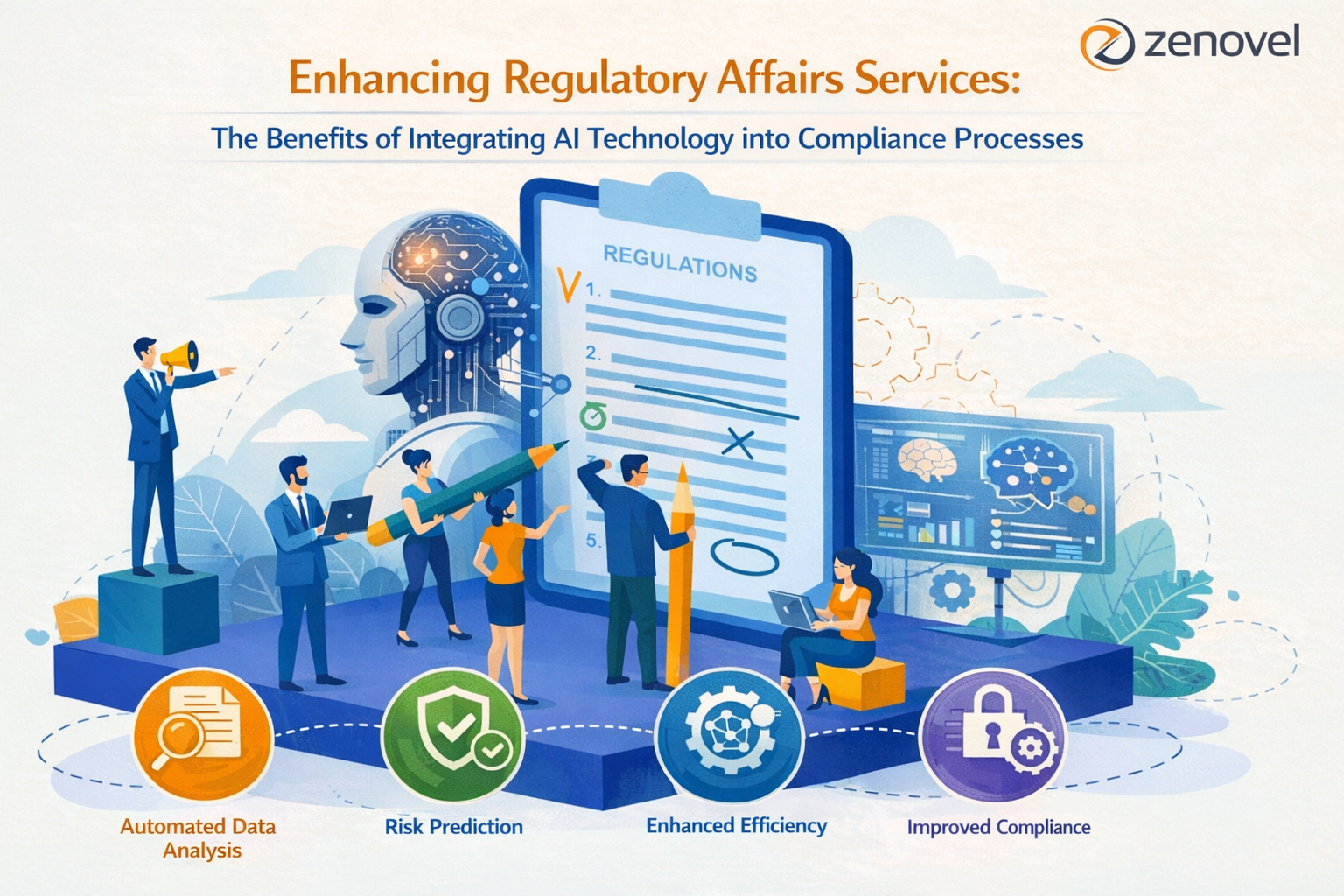
Moving forward into 2026, the pharmaceutical and healthcare industries are embarking on a transformation with artificial intelligence (AI) as a catalyst for change in regulatory affairs. At Zenovel, we see great potential for AI technology to be integrated into compliance efforts to create efficiencies, accuracy, and strategic choices. This blog
We provide affordable, innovative, and high-quality solutions to pharma industry with ethics driven research and increasing accessibility to high quality medicine worldwide
Copyright © 2024 Zenovel. All rights reserved.