
The Role of GxP Services in India in Strengthening Documentation Control for GMP and GCP
In the pharmaceutical and life sciences sectors, documentation control is critical for ensuring compliance, product quality, and patient safety. Adhering
We Embody a Legacy of Incredible Discoveries, Exploring the Boundaries of Innovation and Science

At Zenovel, we integrate our extensive expertise across a wide range of domains, from Good Clinical Practice to Regulatory Affairs, to present valuable insights and thought leadership. Each blog reflects our intense dedication to innovation, offering a forum to share our perspectives, groundbreaking ideas, and industry expertise. Through our blogs, we hope to uplift, inform, and support scientific, medical, and pharmaceutical innovation.

In the pharmaceutical and life sciences sectors, documentation control is critical for ensuring compliance, product quality, and patient safety. Adhering

India’s pharmaceutical industry is poised for significant growth, with projections estimating its market value to reach $130 billion by 2030.
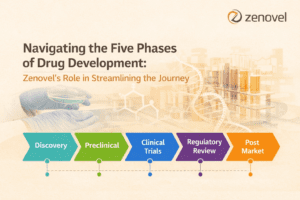
Developing new medications is a complex and costly scientific process that takes 10-15 years and requires billions of dollars to

Risk Based Monitoring (RBM) is a more efficient alternative to traditional clinical trials, focusing on high-risk areas through data-driven insights.

Welcome to the Zenovel Blog! We are a leading provider of drug regulatory consulting and education, focusing on advancing pharmaceutical

The international pharmaceutical landscape is inherently connected to the global economy and global trade, which is consisted with the quality

Drug discovery to market approval is a lengthy and arduous endeavor, lasting a year or more while taking an enormous amount of resources and finances. Among the various processes in going from discovery to market, Chemistry, Manufacturing, and Controls (CMC) is essential in establishing that drugs are safe and effective

Clinical trials are obviously the spine of medical innovation, but their success defines through constant and stringent oversight process compliance to regulatory requirements, patient safety, and data integrity. Zenovel Pharma Services LLP, an experienced global leader in providing clinical research support, is committed to providing a level of sponsor oversight

Pharmaceutical and medical device companies face a significant challenge in navigating the complex regulatory landscapes of the European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA). Consulting services provided by Zenovel offer strategic guidance, hands-on support, and deep regulatory knowledge to help companies pass EMA and FDA

Clinical trials play a vital role in the development of new medications, ensuring they are safe and effective before reaching the market. Throughout the drug development process, treatments progress through various trial phases, each providing deeper insights into their potential benefits for patients. Effective clinical trial management (CTM) is critical

The European Union’s Good Manufacturing Practice (GMP) standards, as outlined in the Annex to the Guide to Good Manufacturing Practice for Medicinal Products, ensure the quality and safety of medicinal products, including those used in clinical trials. The Qualified Person (QP) plays a crucial role in batch certification, particularly for

In the intricate process of developing new pharmaceuticals, guaranteeing product safety, effectiveness, and adherence to global regulations is essential. Batch release testing acts as a pivotal quality assurance measure, confirming that medications comply with rigorous criteria before distribution. When combined with International Council for Harmonisation (ICH) standards, this testing framework
We provide affordable, innovative, and high-quality solutions to pharma industry with ethics driven research and increasing accessibility to high quality medicine worldwide
Copyright © 2024 Zenovel. All rights reserved.