
In pharmaceutical development, the acronyms GMP, GCP, and GLP represent the “Golden Trio” of quality standards, ensuring that every drug is safe, effective, and manufactured to high standards. Each pillar governs a specific phase of the product lifecycle, aimed at protecting patients through rigorous quality and compliance.
Table of Contents
ToggleThe Three Pillars: At a Glance
| Pillar | Phase of Drug Lifecycle | Primary Focus |
| Good Laboratory Practice | Preclinical (Non-Clinical) | Safety of the drug candidate through laboratory studies |
| Good Clinical Practice | Clinical Development | Rights, safety, and well-being of human subjects; data integrity |
| Good Manufacturing Practice | | Manufacturing & Commercial |
-
GLP: Good Laboratory Practice – The Foundation
What It Is:
GLP is a quality system focused on the processes and conditions for planning, performing, monitoring, recording, archiving, and reporting non-clinical health and environmental safety studies.
Why It Matters:

When It Applies:
From the initiation of preclinical testing to the submission of an Investigational New Drug (IND) application or Clinical Trial Application (CTA), the process outlines the early stages of drug development.
GCP: Good Clinical Practice – The Human Element
What It Is:
GCP is an international standard for ethical and scientific quality in the design, conduct, recording, and reporting of clinical trials involving human subjects.
Why It Matters:

When It Applies:
Throughout all phases of clinical development (Phase I through Phase IV).
GMP: Good Manufacturing Practice – The Product Guarantee
What It Is:
GMP is a component of quality assurance that guarantees products are consistently produced and controlled in accordance with quality standards suitable for their intended use.
Why It Matters:

When It Applies:
From clinical trial material manufacturing through commercial production.
How the Golden Trio Works Together
The three pillars are not silos—they are interconnected stages of a seamless quality journey:
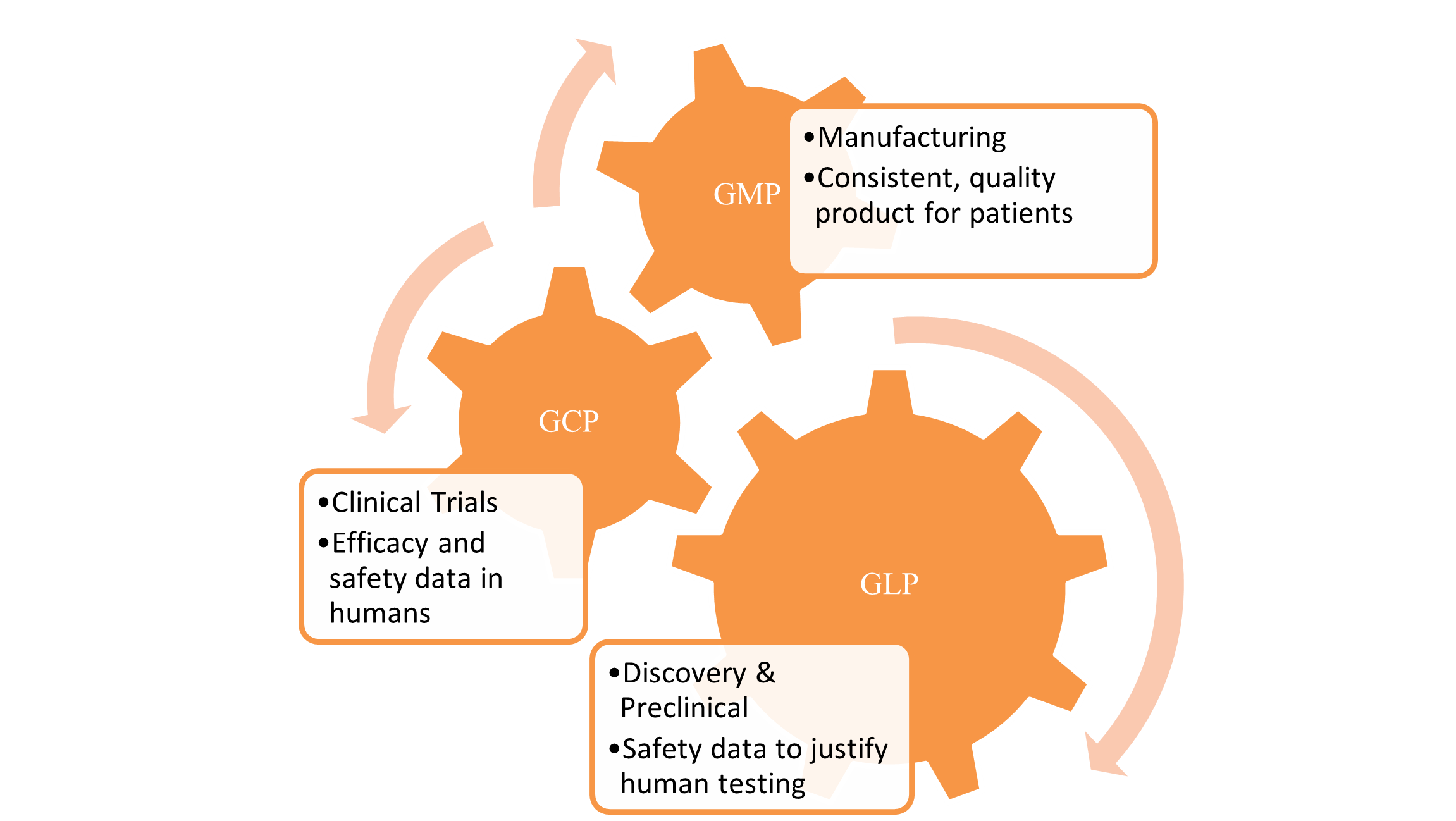
A weakness in any pillar of a development program can lead to significant issues: GLP failures result in no IND, GCP failures lead to rejected clinical data, and GMP failures can cause a lack of product availability or even patient harm.
Consequences of Non-Compliance
Figure 1: Consequences of Non-Compliance

Zenovel: Your Partner Across the Golden Trio
Zenovel offers comprehensive GxP services that cover GLP, GCP, and GMP requirements, backed by extensive regulatory expertise and experience with national and global clients. For more information on their services, visit www.zenovel.com.
Ready to Strengthen Your GxP Compliance?
Contact Zenovel to explore their GLP, GCP, and GMP services, ensuring your product’s transition from lab to patient is grounded in quality and compliance.