
Health Canada’s Good Manufacturing Practices Guide for Drug Products (GUI-0001) is essential for pharmaceutical companies operating in or exporting to Canada. It interprets GMP requirements from Part C, Division 2 of the Food and Drug Regulations, outlining compliance standards for fabricators, packagers, labellers, testers, importers, distributors, and wholesalers.
Understanding GUI-0001 is essential for acquiring and retaining a Drug Establishment Licence (DEL) and for ensuring compliance with Canadian safety and quality standards for products.
Table of Contents
ToggleWhat is GUI-0001?
GUI-0001 is Health Canada’s detailed interpretation of Good Manufacturing Practice (GMP) requirements for drug products, first published in its current version in 2020 to replace the 2018 edition. It acts as the main reference source for compliance in this area.
Domestic manufacturers aiming to align with Canadian regulations must ensure compliance with the relevant legal requirements.
- Importers are accountable for products sourced from overseas facilities.
- Distributors and wholesalers are entities that manage the supply chain of drug products, ensuring their effective distribution to various points of sale.
- Testing laboratories are responsible for conducting release and stability testing to ensure the quality and efficacy of products before they enter the market.
The guide covers GMP requirements for both finished dosage forms and active pharmaceutical ingredients (APIs), incorporating regulatory amendments relevant to API manufacturers.
Key Principles Addressed in GUI-0001
- Quality Management System
GUI-0001 stresses the importance of integrating quality into all operational aspects, highlighting it as a fundamental requirement.

- Quality Control Department
Section C.02.015 of the Food and Drug Regulations pertains to the Quality Control Department, identifying quality control as a significant deficiency in Health Canada inspections, which is noted as the largest deficiency area. Expectations for quality control processes are set within this context.

- Data Integrity
The 2017 draft updates to GUI-0001 introduced requirements for data integrity, aligning with

- Manufacturing Control
The Manufacturing Control section (C.02.019) regulates the entire process from raw materials to finished product release, encompassing essential components throughout this workflow.

- Finished Product Testing Requirements
GUI-0001 delineates specific responsibilities for importers, noting that amendments from July 2020, which followed the CUSMA implementation, allow certain lower-risk non-prescription drug products to potentially meet reduced testing requirements.

- Facility and Equipment Requirements
The guide provides detailed expectations for:

- Documentation and Record-Keeping
Comprehensive documentation is essential for GMP compliance, as outlined in GUI-0001.

- Stability Program
Drug products must demonstrate stability throughout their shelf life.

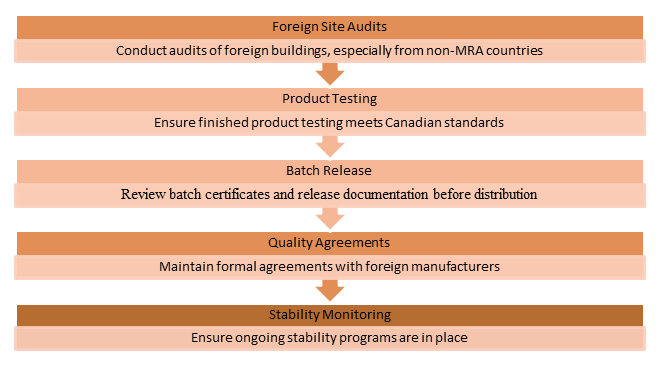
- Importer Responsibilities Under GUI-0001
For companies importing drug products into Canada, GUI-0001 imposes significant obligations on the Canadian licence holder.
- Inspection and Enforcement
Health Canada’s Regulatory Operations and Enforcement Branch (ROEB) performs Good Manufacturing Practice (GMP) inspections to ensure compliance with GUI-0001.
The Drug and Health Products Inspections Database (DHPID) offers public access to inspection results, enhancing transparency and regulatory oversight. Common deficiencies noted include inadequate change control systems, quality control department shortcomings, manufacturing control failures, and documentation gaps.

GUI-0001 vs. GUI-0158: Understanding the Difference
GUI-0001 and GUI-0158, which governs Natural Health Products (NHPs), both address GMP but apply to different product categories and possess distinct requirements. Notably, GUI-0158 was updated in September 2025, with compliance mandated by March 2026.
Zenovel offers comprehensive GMP services in accordance with Health Canada’s GUI-0001. With abundant years of regulatory expertise and experience serving clients locally and globally, our assists pharmaceutical companies in achieving Canadian GMP compliance. We offer complete set of GMP services from Gap analysis, inspection readiness , quality system implementation to documentation remediation. For insights and details visit our website on www.zenovel.com or request your proposal via bd@zenovel.com
We are ready to hear from you soon!!!