
Pharmaceutical startups encounter a challenging journey from laboratory breakthroughs to marketable products, facing significant scientific and regulatory hurdles. A sound global regulatory strategy is crucial, as it can expedite approval processes or lead to delays and failures. Startups, with their limited resources and tight timelines, need to make early strategic decisions that significantly influence their development path, in contrast to established companies with dedicated regulatory teams.
Zenovel facilitates pharmaceutical startups by leveraging abundant years of regulatory expertise and serving over clients nationally and globally, transforming regulatory challenges into competitive advantages.
Why Startups Need a Dedicated Regulatory Strategy
The regulatory landscape of 2026 presents both significant opportunities and challenges for emerging pharmaceutical companies, driven by the FDA’s move toward single-trial approvals, the proposed EU Biotech Act, and India’s regulatory sandbox initiatives, all of which are transforming global development strategies for startups.
Price of Failure:
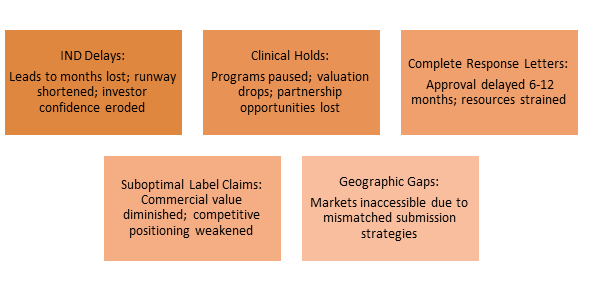
The success of startups hinges on integrating regulatory thinking as a fundamental strategic function from the outset, rather than considering it as an afterthought.
Phase 1: Pre-IND Strategy – Setting the Stage
- Target Product Profile (TPP) Development
Every regulatory strategy relies on the Target Product Profile (TPP), which outlines the intended indication, patient population, labeling claims, competitive positioning, unmet medical needs, and prioritized global markets. The TPP serves as a guiding framework for all regulatory decisions, ensuring that clinical trials, CMC development, and regulatory interactions are aligned with it.
- Regulatory Pathway Selection
Startups should carefully consider their options as not all pathways to success are the same.
| Pathway | Applicable for | Main considerations |
| 505(b)(1) NDA | New chemical entities | Full safety/efficacy data required |
| 505(b)(2) | Modified versions; new indications | Reliance on literature or prior findings |
| BLA | Biologics | Extensive CMC focus; comparability challenges |
| Biosimilar | Follow-on biologics | | Stepwise approach; extensive analytical similarity |
- Early Regulatory Engagement
The FDA’s INTERACT meetings, Pre-IND meetings, and EMA’s innovation task forces provide startups essential chances to coordinate with regulators prior to significant investments, aiding in the clarification of nonclinical package requirements, confirmation of clinical design acceptability, identification of potential issues, and relationship-building with review divisions.
- Expedited Program Assessment
Startups are encouraged to assess their eligibility for expedited programs at an early stage.
| Agency | Program | Benefit |
| FDA | Fast Track | Rolling review; frequent interactions |
| FDA | Breakthrough Therapy | Intensive guidance; organizational commitment |
| EMA | PRIME | Early support; accelerated assessment |
| Japan (PMDA) | Sakigake | Priority consultation; fast-track review |
| FDA | Regenerative Medicine Advanced Therapy (RMAT) | For cell/gene therapies; accelerated pathways |
- India’s Regulatory Sandbox Opportunity
India’s Department of Biotechnology is creating regulatory sandboxes for the pharmaceutical sector to expedite product approvals. This initiative enables innovators to test their products under relaxed requirements and fosters early structured interactions with CDSCO, providing startups with quicker market entry and essential regulatory feedback.
Phase 2: IND/CTA Submission – Initiating Human Trials
- Global Submission Strategy
For startups aiming for global development, the choice of initial filing location is crucial, influenced by factors such as regulatory predictability, access to patient populations, intellectual property issues, cost, infrastructure needs, and possibilities for expedited pathways. Australia’s TGA provides a strategic advantage with its recent ICH E6(R3) GCP implementation, offering a 12-month transition period for compliance with either E6(R2) or E6(R3), which startups can utilize.
- CMC Readiness for Early Trials
Manufacturing complexity is crucial for biologics and cell therapies. Startups should focus on CMC planning early and collaborate with experienced CDMOs that provide scalable, GMP-ready processes to prevent expensive redesigns later.
- IND Package Components
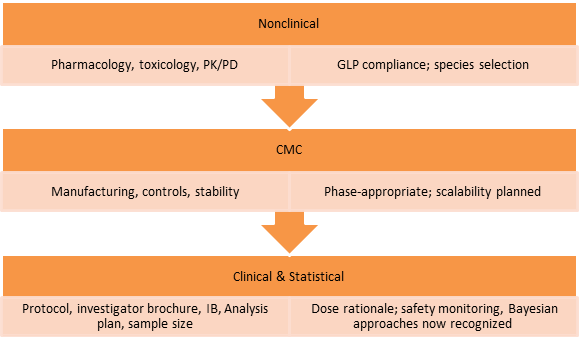
Phase 3: Clinical Development – Offering the Evidence Base
- Adaptive and Innovative Trial Designs
The FDA’s January 2026 draft guidance on Bayesian methodology presents opportunities for startups by allowing the incorporation of prior information, adaptable trial designs, smaller sample sizes, and more efficient dose-finding. Concurrently, China’s NMPA released guidance supporting the use of Bayesian external information borrowing methods in clinical research.
- ICH E6(R3) Implementation
As of January 13, 2026, Australia’s TGA has implemented ICH E6(R3) GCP, focusing on risk-based approaches for critical data, essential records through eTMF/CTMS, proactive oversight, and documented training for personnel. A 12-month transition period is provided for startups to update their quality systems and training programs.
- The EU Biotech Act:
The proposed EU Biotech Act, announced in December 2025, promises significant changes for clinical trial sponsors.
| Change | Current | Proposed |
| Multinational trial authorization | 106 days | 75 days |
| No-information-request authorization | 75 days | 47 days |
| SPC extension | Standard | +12 months for EU-developed products |
| ATMP review | Additional 50 days | Removed |
The Act introduces single core dossiers for investigational medicinal products, allowing one dossier to support multiple clinical trials, which significantly enhances efficiency for startups engaged in iterative development programs.
- Combination Studies with Diagnostics
For startups creating products with companion diagnostics, the EU Biotech Act suggests a unified assessment process for combined studies of IMPs and medical devices/in vitro diagnostics, resolving issues in existing fragmented frameworks.
Phase 4: NDA/MAA Submission –The Way to Approval
Rolling review enables products with Fast Track or Breakthrough designation to submit completed sections of their dossier as they are ready, rather than waiting for the entire submission. This process can reduce review timelines by several months.
- The Single Pivotal Trial Era
FDA’s February 2026 announcement established that one adequate and well-controlled study, supplemented by confirmatory evidence, will be the standard for marketing authorization. This shift reflects that approximately 60% of first-of-a-kind drugs have already received single-trial approvals in the last five years.
For startups, this emphasizes the need to focus on creating compelling confirmatory evidence, strategically utilizing real-world data and external controls, and enhancing mechanistic understanding and translational data.

Phase 5: Post-Approval – Lifecycle Management
- Post-Approval Commitments
Regulatory approvals for startups generally require additional commitments such as post-marketing studies or clinical trials, necessitating careful financial and operational planning.

- The US-India Collaboration Model
Startups aiming for global reach can benefit from the US-India collaboration, which combines US strengths in capital, innovation, regulatory frameworks, and execution with India’s cost-efficient R&D, scalable clinical infrastructure, competitive manufacturing, and regulatory strategy for global compliance.
This approach effectively decreases development burn rates and ensures regulatory credibility, leveraging India’s extensive patient base for oncology trials and its manufacturing capabilities for large-scale production.
Zenovel offers comprehensive regulatory consulting and strategic services designed specifically for pharmaceutical startups. Our services cover various development phases, including pre-IND strategy, IND/CTA submission, clinical development, NDA/MAA submission, and post-approval activities. Key services include target product profile development, regulatory pathway selection, dossier preparation, protocol design, and lifecycle management. Additionally, Zenovel provides specialized expertise in orphan drug applications, accelerated program requests, and biosimilar development, supporting emerging companies in navigating the complexities of drug development from IND to approval.
Ready to Accelerate Your Regulatory Journey? Contact Zenovel today to discuss how our regulatory consulting and strategic services can guide your startup from discovery through global approval.
References:
- BioPharma Shakti 2026 must translate into predictable regulatory timelines: Venkat Nelabhotla
- Global Regulatory Updates on Clinical Trials (January 2026)
- Key Manufacturing Actions for ATMPs After 2024–2025 Regulatory Shifts