
The year 2026 represents a pivotal moment for artificial intelligence in clinical research, with generative AI transitioning from a novel concept to an operational essential. This technology, capable of drafting reports and creating synthetic control arms, offers the potential to shorten timelines, cut costs, and extract valuable insights from unstructured data. However, it also introduces significant regulatory and ethical challenges, including algorithm validation, accountability for errors in AI-generated content, and the safeguarding of patient privacy amid extensive training datasets.
As generative AI is integrated into clinical trial operations, the industry faces the challenge of balancing innovation with ethical research standards of safety, integrity, and human-centeredness. Zenovel is poised to assist sponsors in the responsible integration of generative AI tools, maximizing opportunities while mitigating regulatory risks.
-
Table of Contents
ToggleGenerative AI in Clinical Research:
Generative AI encompasses algorithms that generate new content, including text, images, code, and structured data, derived from patterns in training data. In clinical research, this potential is significant.
Generative AI distinguishes itself from traditional AI by its ability to create rather than merely classify or predict, presenting both significant capabilities and validation challenges.
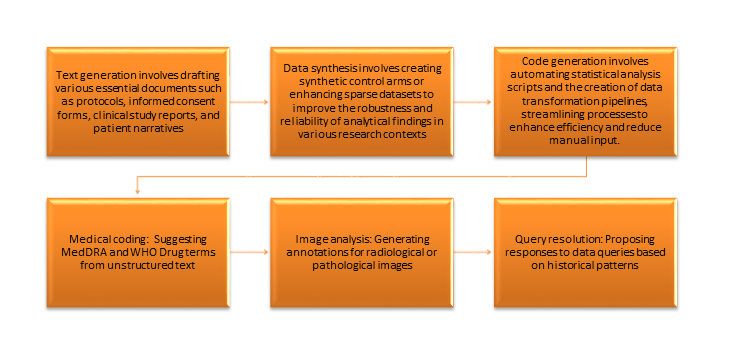
Opportunities: Where Generative AI Delivers Value
-
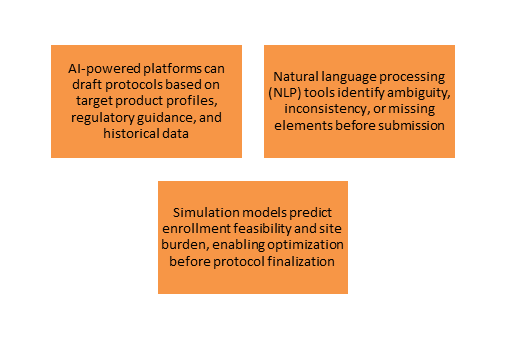
Protocol Development and Optimization
-
- The Challenge: Protocol design is a resource-intensive, iterative process that frequently results in amendments, causing trial delays of several months.
- Generative AI Opportunity
- Effect: Organizations utilizing generative AI for protocol development have observed reductions in protocol amendment rates by 20-30% and expedited regulatory submission timelines by weeks.
👉 Accelerate Smarter Trial Design
Reduce protocol amendments and improve study efficiency with AI-ready clinical operations support from Zenovel.
-
Patient Recruitment and Engagement
-
- The Challenge: Over 50% of clinical trials face enrollment delays, primarily attributed to mismatched eligibility criteria and ineffective recruitment messaging.
- Generative AI Opportunity:
- Effect: Several large CROs utilize generative AI chatbots, reducing patient screening time by 40-60% and enhancing enrollment diversity.
👉 Improve Patient Enrollment with AI-Driven Strategies
Enhance recruitment speed, diversity, and patient engagement through compliant AI-enabled clinical workflows.

-
Clinical Data Management and Cleaning
- The Challenge: Manual data cleaning accounts for 30% of data management efforts, frequently causing delays of weeks or even months for queries.
- Generative AI Opportunity:
-
- Effect: Generative AI can decrease manual data management efforts by 30-50%, while enhancing consistency and lowering query volume.
👉 Simplify Clinical Data Management
Leverage intelligent data review and AI-supported quality processes to reduce delays and improve consistency.
-
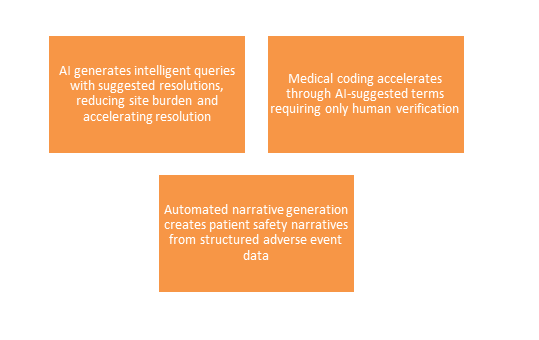
Regulatory Document Generation
- The Challenge: Clinical study reports, investigator brochures, and submission dossiers necessitate significant medical writing effort and extensive cross-functional collaboration over several weeks.
- Generative AI Opportunity
-
- Effect: Regulators have not formally endorsed AI-generated submissions, but they do accept AI-assisted content if there is documented human oversight and verification.
👉 Faster Regulatory Documentation Starts Here
Support your submission readiness with compliant AI-assisted medical writing and regulatory expertise.

-
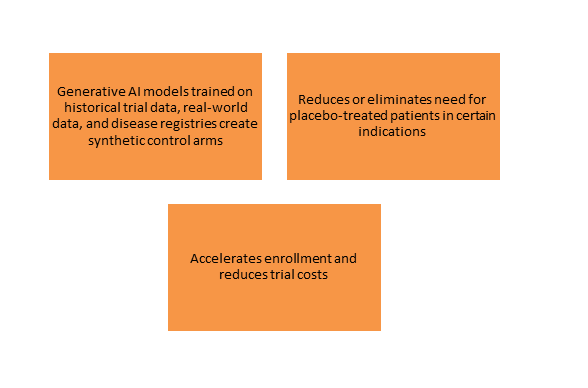
Synthetic Control Arms
- The Challenge: Recruiting control arms in rare diseases or ethical concerns about placebo use may delay or impede clinical trials.
- Generative AI Opportunity:
-
- Regulatory Recognition: The FDA has recognized the use of synthetic control arms in certain situations, especially when historical data is reliable and the disease is well-defined, but justifications must be provided on a case-by-case basis.
👉 Explore AI-Powered Trial Innovation
Discover how synthetic control strategies can support rare disease and complex clinical studies responsibly.
-
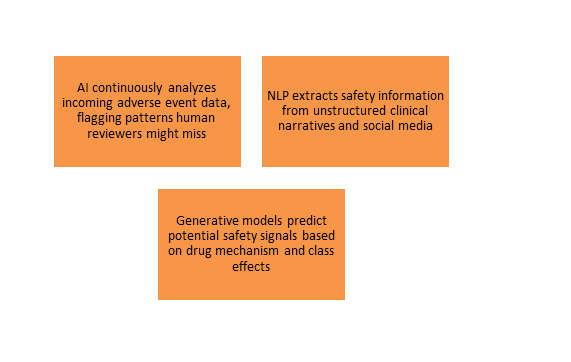
Safety Monitoring and Signal Detection
- The Challenge: Traditional safety monitoring is based on periodic data reviews, which may lead to delays in signal identification.
- Generative AI Opportunity:
-
- Effect: AI-enhanced safety monitoring allows for real-time signal detection, enabling the identification of safety concerns significantly earlier than traditional periodic reviews.
👉 Enhance Pharmacovigilance with AI Insights
Enable faster safety signal identification while maintaining compliance and human oversight.
-
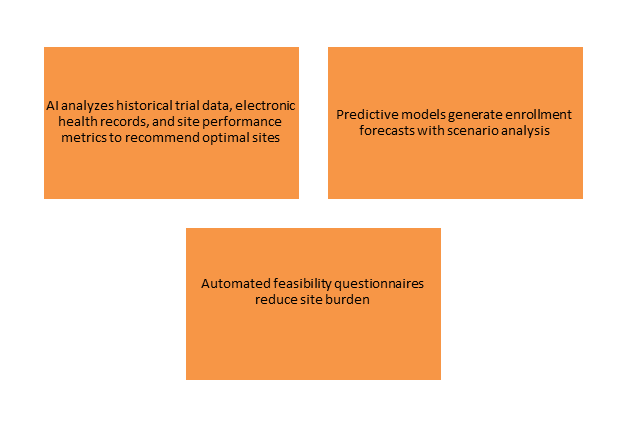
Site Selection and Feasibility
- The Challenge: Identifying suitable sites for patient populations with the necessary experience and capacity is a major bottleneck.
- Generative AI Opportunity
-
- Effect: Real world platforms have demonstrated ability to identify 80% of needed sites in six weeks for global Phase III programs .
👉 Find the Right Sites Faster
Use data-driven feasibility strategies to identify high-performing clinical trial sites globally.
Regulatory Challenges: The Other Side of the Coin
-
Algorithm Validation and Transparency
- The Challenge: Generative AI models, especially large language models, operate as “black boxes,” with opaque and difficult-to-audit internal decision-making processes.
- Regulatory Expectations:
-
- Emerging Consensus: Regulators require sponsors to validate AI systems, encompassing user requirements, design specifications, testing documentation, and continuous monitoring. For generative AI, this validation must ensure that the outputs are accurate, unbiased, and reproducible.
👉 Build Trustworthy AI Clinical Systems
Ensure your AI tools align with evolving regulatory expectations for validation, transparency, and oversight.

-
Data Privacy and Security
- The Challenge: Generative AI models trained on patient-level data may inadvertently memorize and reproduce identifiable information.
- Regulatory Framework:
-
- Best Practices: To ensure compliance and privacy, use synthetic or anonymized data for model training, implement technical controls to prevent reproduction of patient-identifiable information, and establish data processing agreements with AI vendors focusing on security and privacy obligations.
👉 Protect Sensitive Clinical Data
Implement privacy-first AI frameworks that support compliance, security, and patient trust.

-
Accountability and Liability
- The Challenge: When AI generates content such as patient narratives, safety signals, or protocol sections, questions arise regarding accountability for potential errors in the generated output.
- Regulatory Framework
- Current Approach: Regulators mandate that qualified individuals review and approve all AI-generated content for regulatory submissions or patient safety, underscoring that AI is intended to assist, not replace human expertise.

-
Bias and Fairness
- The Challenge: Generative AI models using historical data can reinforce or enhance biases in trial enrollment, representation, and clinical decision-making.
- Regulatory Scrutiny
-
- Emerging Guidance: Sponsors must conduct bias assessments on AI tools in clinical trials, analyzing the composition of training data and testing outputs across diverse patient populations.
👉 Promote Ethical and Inclusive Clinical Research
Reduce bias risks in AI-driven clinical trials with expert oversight and responsible implementation practices.

-
Data Integrity and Audit Trails
- The Challenge: Generative AI produces new data, which poses a challenge to conventional data integrity frameworks that rely on traceable transformations.
- Regulatory Framework:
- Emerging Practices: Sponsors using generative AI are creating AI usage logs to record the instances of AI application, including details about the inputs received, outputs produced, and any human review conducted.

-
Intellectual Property and Ownership
- The Challenge: Generative AI models that utilize copyrighted material for training give rise to concerns regarding the ownership of the outputs they produce.
- Regulatory Considerations: IP issues indirectly influence sponsors’ ability to operate and publish freely.
- Current Position: Regulators prioritize the accuracy and integrity of AI-generated content, but sponsors must ensure that such content does not use proprietary information without proper authorization.
Zenovel offers support to sponsors in responsibly utilizing generative AI, with a team that possesses deep clinical, regulatory, and technical expertise to ensure the integration of AI enhances trial quality and compliance.
👉 Ready to Responsibly Integrate Generative AI?
Zenovel helps sponsors adopt generative AI in clinical trials while maintaining regulatory compliance, data integrity, and patient safety.
Ready to Explore Generative AI in Your Trials? Contact Zenovel to explore how our expertise in AI-ready clinical operations and regulatory matters can assist you in effectively utilizing generative AI.
