
Clinical trials are becoming increasingly complex as pharmaceutical, biotechnology, and medical device companies expand research programs globally. In Brazil, sponsors must comply with strict regulatory standards, ethical guidelines, and operational requirements to ensure successful clinical trial execution. This is where comprehensive Good Clinical Practice (GCP) services in Brazil play a critical role.
Zenovel provides end-to-end GCP compliance services, helping sponsors maintain regulatory alignment, patient safety, data integrity, and operational efficiency throughout every stage of clinical development. With expertise in clinical operations, quality assurance, regulatory affairs, pharmacovigilance, and clinical monitoring, Zenovel supports compliant and inspection-ready clinical trials across Brazil.
Table of Contents
ToggleIntroduction to GCP-Compliant Clinical Trials in Brazil
Brazil has become an important destination for global clinical research due to its large patient population, experienced investigators, and expanding healthcare infrastructure. However, conducting clinical trials in Brazil requires adherence to international ICH-GCP guidelines, local regulatory requirements, and ANVISA compliance standards.
Sponsors conducting Phase I–IV studies often require specialized Clinical Research Organization (CRO) services in Brazil to manage clinical trial compliance, reduce operational risks, and maintain study timelines.
Zenovel supports pharmaceutical companies, biotech organizations, medical device manufacturers, and research sponsors through integrated clinical trial compliance services in Brazil designed to improve study quality and regulatory readiness.
Understanding Good Clinical Practice (GCP) in Clinical Research
Good Clinical Practice (GCP) is an internationally recognized quality standard that governs the design, conduct, monitoring, recording, and reporting of clinical trials involving human participants.
The primary goals of GCP compliance include:
- Protecting participant safety and rights
- Ensuring ethical clinical research practices
- Maintaining accurate and reliable clinical data
- Supporting regulatory approval readiness
- Improving transparency and documentation standards
Strong GCP compliance services help organizations minimize protocol deviations, reduce compliance risks, and maintain inspection readiness throughout the study lifecycle.
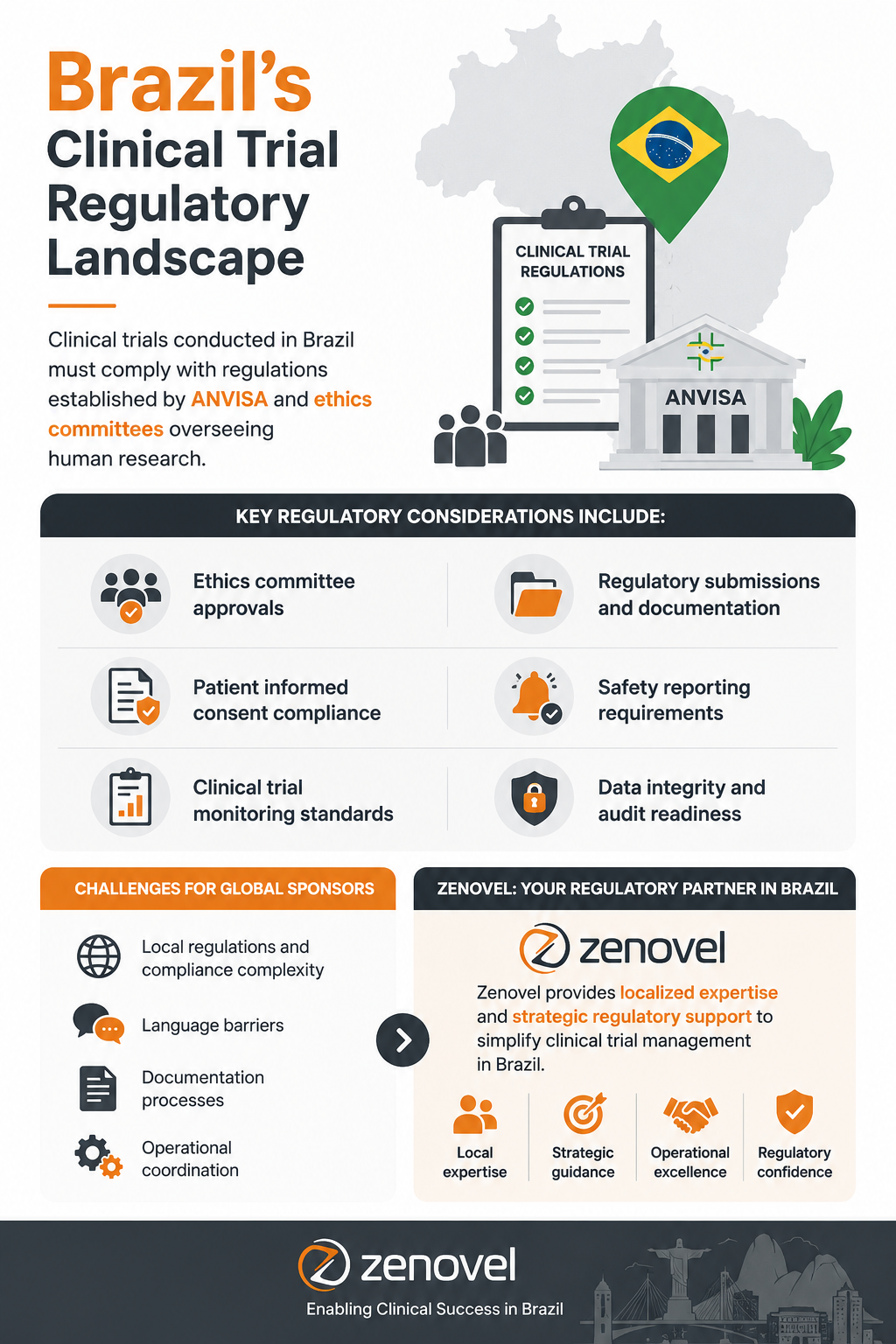
Brazil’s Clinical Trial Regulatory Landscape
Clinical trials conducted in Brazil must comply with regulations established by ANVISA and ethics committees overseeing human research.
Key regulatory considerations include:
- Ethics committee approvals
- Regulatory submissions and documentation
- Patient informed consent compliance
- Safety reporting requirements
- Clinical trial monitoring standards
- Data integrity and audit readiness
Global sponsors entering Brazil often face challenges related to local regulations, language barriers, documentation processes, and operational coordination. Zenovel provides localized expertise and strategic regulatory support to simplify clinical trial management in Brazil.
Common GCP Compliance Challenges in Brazil
Maintaining GCP compliance during a clinical trial requires continuous oversight and quality management. Sponsors frequently encounter challenges such as:
Site Management Complexity
Managing multiple clinical trial sites while ensuring consistent protocol adherence can be difficult.
Regulatory Documentation Gaps
Incomplete or inconsistent documentation may delay approvals and inspections.
Patient Recruitment and Retention
Recruiting eligible participants while maintaining ethical standards remains a key operational challenge.
Data Integrity Risks
Accurate clinical data collection, monitoring, and reporting are essential for successful submissions.
Inspection Readiness
Preparing for regulatory inspections requires strong quality systems and audit-ready documentation.
Zenovel addresses these challenges through proactive monitoring, risk-based quality management, and comprehensive clinical operations support.
How Zenovel Supports GCP-Compliant Clinical Trials in Brazil
Zenovel delivers integrated GCP services in Brazil that support efficient, compliant, and high-quality clinical trial execution.
Clinical Monitoring Services
Zenovel provides comprehensive clinical trial monitoring services in Brazil to ensure study activities align with approved protocols, GCP standards, and regulatory expectations.
Monitoring activities include:
- Site initiation visits
- Routine monitoring visits
- Source data verification (SDV)
- Site closeout activities
- Protocol compliance assessments
- Investigator support and training
These services help sponsors maintain trial quality, reduce protocol deviations, and improve data accuracy.
Risk-Based Monitoring Strategies
Modern clinical trials require efficient oversight models that prioritize critical risk areas.
Zenovel implements risk-based monitoring (RBM) services to:
- Identify high-risk study activities
- Improve monitoring efficiency
- Enhance patient safety oversight
- Reduce operational costs
- Strengthen compliance management
Risk-based monitoring supports scalable and data-driven clinical trial operations across Brazil.
Clinical Site Management Support
Effective site management is essential for maintaining study timelines and regulatory compliance.
Zenovel’s clinical site management services include:
- Site feasibility assessments
- Investigator coordination
- Site activation support
- Staff training and GCP education
- Patient recruitment assistance
- Site performance tracking
This structured approach helps improve site productivity and study execution quality.
GCP Audits and Quality Assurance Services
Quality assurance is critical for maintaining inspection readiness throughout the clinical trial lifecycle.
Zenovel offers specialized GCP audit services in Brazil including:
- Internal quality audits
- Site audits
- Vendor qualification audits
- Trial Master File (TMF) reviews
- CAPA management support
- Inspection readiness assessments
These services help organizations identify compliance gaps early and strengthen operational quality systems.
Regulatory Affairs Support for Clinical Trials
Navigating Brazil’s regulatory environment requires local expertise and strategic planning.
Zenovel provides regulatory affairs services in Brazil including:
- Clinical trial application support
- Regulatory documentation preparation
- Ethics committee coordination
- Submission management
- Regulatory gap analysis
- Compliance strategy development
This support accelerates regulatory processes while reducing submission-related delays.
Pharmacovigilance and Safety Monitoring
Patient safety remains a central component of GCP-compliant clinical trials.
Zenovel’s pharmacovigilance services support:
- Adverse event reporting
- Serious adverse event (SAE) management
- Safety data review
- Signal detection and risk management
- Safety database support
- Regulatory safety reporting compliance
These services help sponsors maintain global patient safety standards and regulatory obligations.
Clinical Data Management and Data Integrity
Accurate clinical data is essential for regulatory approvals and study success.
Zenovel delivers compliant clinical data management services focused on:
- Electronic data capture (EDC) management
- Data validation and cleaning
- Query resolution
- Database review
- Audit trail maintenance
- Data integrity compliance
Strong data management processes improve study reliability and inspection readiness.
Technology-Driven GCP Compliance Solutions
Zenovel integrates modern technologies to improve operational efficiency and compliance oversight.
Technology-enabled solutions include:
- Clinical Trial Management Systems (CTMS)
- Electronic Trial Master File (eTMF) systems
- Risk-based quality management tools
- Remote monitoring capabilities
- Compliance tracking dashboards
- Secure clinical data systems
These technologies support faster decision-making and enhanced clinical trial visibility.
Benefits of Partnering with Zenovel for Clinical Trials in Brazil
Sponsors partnering with Zenovel benefit from:
Local Regulatory Expertise
Deep understanding of Brazilian clinical trial regulations and operational requirements.
End-to-End CRO Support
Integrated clinical development, monitoring, quality assurance, and regulatory services.
Improved Compliance Management
Proactive strategies to maintain GCP compliance throughout study execution.
Faster Trial Execution
Efficient workflows and experienced clinical operations teams accelerate timelines.
Inspection Readiness
Audit-focused processes support successful regulatory inspections and submissions.
Cost-Effective Clinical Trial Support
Scalable solutions designed for emerging biotech companies and global pharmaceutical organizations.
Why Brazil is a Strategic Location for Clinical Trials
Brazil continues to attract global clinical research investments due to:
- Large and diverse patient populations
- Experienced clinical investigators
- Expanding research infrastructure
- Strong therapeutic area expertise
- Opportunities for faster patient enrollment
- Increasing international collaboration in clinical research
These factors make Brazil an important market for global clinical development programs.

Best Practices for Maintaining GCP Compliance
Sponsors can improve trial quality and compliance through:
- Continuous GCP training programs
- Routine quality audits
- Effective documentation management
- Risk-based monitoring implementation
- Strong sponsor-CRO collaboration
- Ongoing regulatory updates and compliance reviews
Maintaining proactive oversight helps reduce operational and regulatory risks.
Future Trends in GCP and Clinical Trial Compliance
The clinical research industry is evolving rapidly with increasing adoption of:
- Decentralized clinical trials (DCTs)
- AI-driven clinical analytics
- Remote monitoring technologies
- Digital patient engagement tools
- Advanced compliance automation systems
Organizations adopting modern compliance strategies will be better positioned for future clinical trial success.
Conclusion
Conducting successful clinical trials in Brazil requires strong regulatory expertise, operational efficiency, and continuous Good Clinical Practice compliance. Sponsors must ensure patient safety, data integrity, ethical conduct, and inspection readiness throughout every stage of the study.
Zenovel provides comprehensive GCP services in Brazil, supporting pharmaceutical, biotech, and medical device companies with integrated clinical trial solutions tailored to global compliance standards. From clinical monitoring and regulatory affairs to quality assurance and pharmacovigilance, Zenovel helps organizations execute compliant, efficient, and high-quality clinical trials in Brazil.
Frequently Asked Questions (FAQs)
What are GCP services in clinical trials?
GCP services help ensure clinical trials are conducted ethically, safely, and in compliance with international regulatory standards.
Why is GCP compliance important in Brazil?
GCP compliance supports patient safety, data integrity, regulatory approvals, and successful clinical trial execution.
What industries benefit from GCP services?
Pharmaceutical companies, biotechnology firms, medical device manufacturers, and clinical research organizations benefit from GCP compliance support.
Does Zenovel provide clinical monitoring services in Brazil?
Yes, Zenovel offers comprehensive clinical monitoring, site management, audit support, and regulatory compliance services across Brazil.
How do GCP audits improve clinical trial quality?
GCP audits help identify compliance gaps, improve documentation quality, strengthen operational processes, and maintain inspection readiness.
