
Table of Contents
ToggleThe Growing Importance of Pharmacokinetics Services in Drug Development
India’s clinical research sector is transitioning towards Pharmacokinetics (PK) services, gaining attention as a vital aspect of drug development by 2026. Once a secondary element, PK analysis now plays a crucial role in regulatory submissions, precision dosing, and biologic development. This shift is catalyzed by a blend of scientific talent, updated regulations, technological investments, and competitive pricing, positioning India as the leading hub for PK solutions globally.
The Rise in PK Demand:
Pharmacokinetics, the analysis of drug ADME (absorption, distribution, metabolism, and excretion), is increasingly vital in drug development, with the global PK testing services market expected to grow from $395 million in 2025 to $651 million by 2032, at a CAGR of 7.5%. This surge is driven by the rise of biologic and biosimilar developments requiring advanced PK analyses, regulatory agencies demanding detailed PK data for drug approvals, and a burgeoning clinical trial landscape with over 480,000 active studies producing extensive PK sampling points, significantly increasing laboratory demand.
For global pharmaceutical companies, pharmacokinetic (PK) analysis is essential and should be integrated as a core strategic capability rather than treated as an afterthought.
div style=”border-left: 5px solid #f59221; padding: 20px; background: #fef9f4; margin: 30px 0; border-radius: 6px;”>
👉 Accelerate Your PK Study Timelines
Get reliable pharmacokinetics support with advanced bioanalytical capabilities and regulatory-ready data from Zenovel.
The Five Pillars of a Global PK Hub in India:
India’s ascent as a PK powerhouse is the result of strategic, long-term investments in five key areas.
- A Booming Market with Unmatched Momentum
The bioanalytical testing service market in India reached $120.6 million in 2025 and is projected to grow to $242.5 million by 2034, with a CAGR of 7.83%. The pharmacokinetics services market is also growing steadily, influenced by increasing demand for personalized medicine and more complex drug development. Overall, India’s pharmaceutical industry is valued at around $60 billion, with the CDMO sector at $12–13 billion, experiencing significant growth due to global demand for complex generics, biologics, and specialized formulations.
👉 Looking for Scalable PK & Bioanalytical Support?
Zenovel helps pharma and biotech companies streamline PK workflows with global-quality standards and cost-effective execution.
- Regulatory Modernization That Cuts Red Tape
In 2026, the Indian government dramatically transformed the approval process for PK and bioequivalence (BA/BE) studies by amending the New Drugs and Clinical Trials (NDCT) Rules. The approval timeline for these studies has been reduced from 90 days to 45 days, and a “prior intimation” mechanism has been introduced for lower-risk BA/BE studies, enabling manufacturers to start work upon acknowledgment. These reforms aim to simplify regulatory processes, reduce approval times, and enhance India’s appeal as a global hub for pharmaceutical research and development.
👉 Faster Approvals Need Faster PK Partners
Partner with Zenovel for compliant PK and BA/BE study support aligned with evolving CDSCO, FDA, and EMA expectations.
- Globally Harmonized Quality Standards
India’s regulatory alignment in PK data, driven by the CDSCO’s harmonization of bioanalytical method validation guidelines with global standards like ICH M10 and WHO TRS, enhances trust in its data. The 2025 draft guidelines, which advocate for orthogonal analytical validation and reduced reliance on in vivo models, align India’s requirements closely with those of the FDA and EMA. Consequently, PK data from Indian laboratories is increasingly accepted by global regulators, facilitating their inclusion in global submission packages without duplicate testing.
👉 Regulatory-Ready PK Data Starts Here
Our validated bioanalytical processes support accurate, globally acceptable pharmacokinetic data for submissions worldwide.
- World-Class Infrastructure and Technology
India’s bioanalytical laboratories have reached global standards in instrumentation and expertise, particularly in PK analysis through LC-MS/MS technology. They are equipped with advanced instruments like API 4000 and 5500 and comprehensive ELISA and ICP-MS capabilities. However, the success of these labs also relies on experienced teams skilled in assay development and validation, ensuring compliance with stringent regulatory standards.
👉 Advanced Technology. Trusted PK Outcomes.
From LC-MS/MS platforms to expert assay validation, Zenovel delivers precision-driven pharmacokinetics solutions..
- Unbeatable Cost Advantage with No Quality Trade-Off
Outsourcing high-end PK and bioanalytical processes to India provides biopharmaceutical companies with significant cost savings while maintaining quality. This combination is particularly appealing for global pharma under margin pressure. India’s function in pharmaceutical outsourcing has shifted from efficient execution to strategic partnership, with expectations of accounting for 8–10% of global CDMO outsourcing by 2033.
Zenovel is distinguished for its specialized focus on pharmacokinetics services in a rapidly growing landscape. Let’s Zenovel’s Comprehensive Pharmacokinetics Services
👉 Reduce Costs Without Compromising Quality
Outsource complex PK studies to Zenovel and gain strategic scientific partnership with measurable value.
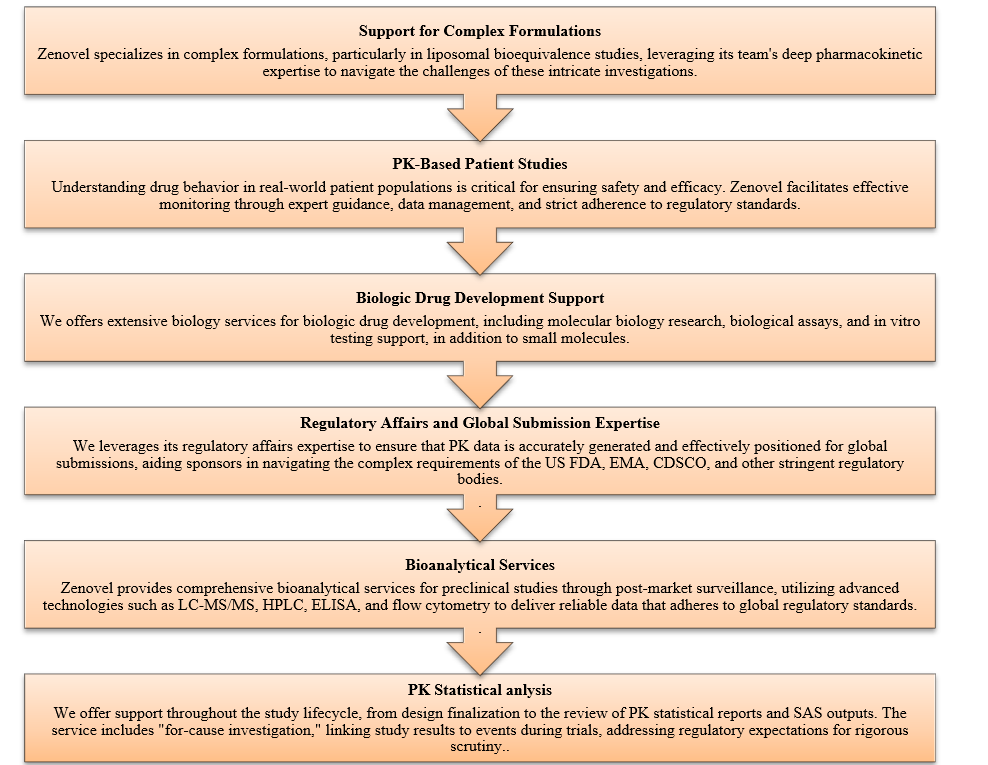
Figure 1: Zenovel PK Support Services
The Road Ahead: India’s PK Future in 2026 and Beyond

👉 Build Your Next Clinical Program with Confidence
Zenovel combines scientific expertise, compliance excellence, and scalable PK services to support modern drug development.
As we look toward the remainder of 2026 and beyond, several trends will continue to accelerate India’s emergence as a global PK hub:
Final Takeaway
Pharmacokinetics has evolved into a strategic discipline crucial for regulatory success, patient safety, and commercial viability in drug development. Leading pharmaceutical companies are increasingly seeking genuine scientific collaboration with India’s pharmacokinetics services, with Zenovel exemplifying expertise, advanced technology, and a commitment to quality.
👉 Need End-to-End Pharmacokinetics Support?
Collaborate with Zenovel for bioanalytical excellence, regulatory confidence, and faster development decisions.
Ready to power your next clinical development program with world-class pharmacokinetics support?
Contact us today on bd@zenovel.com
